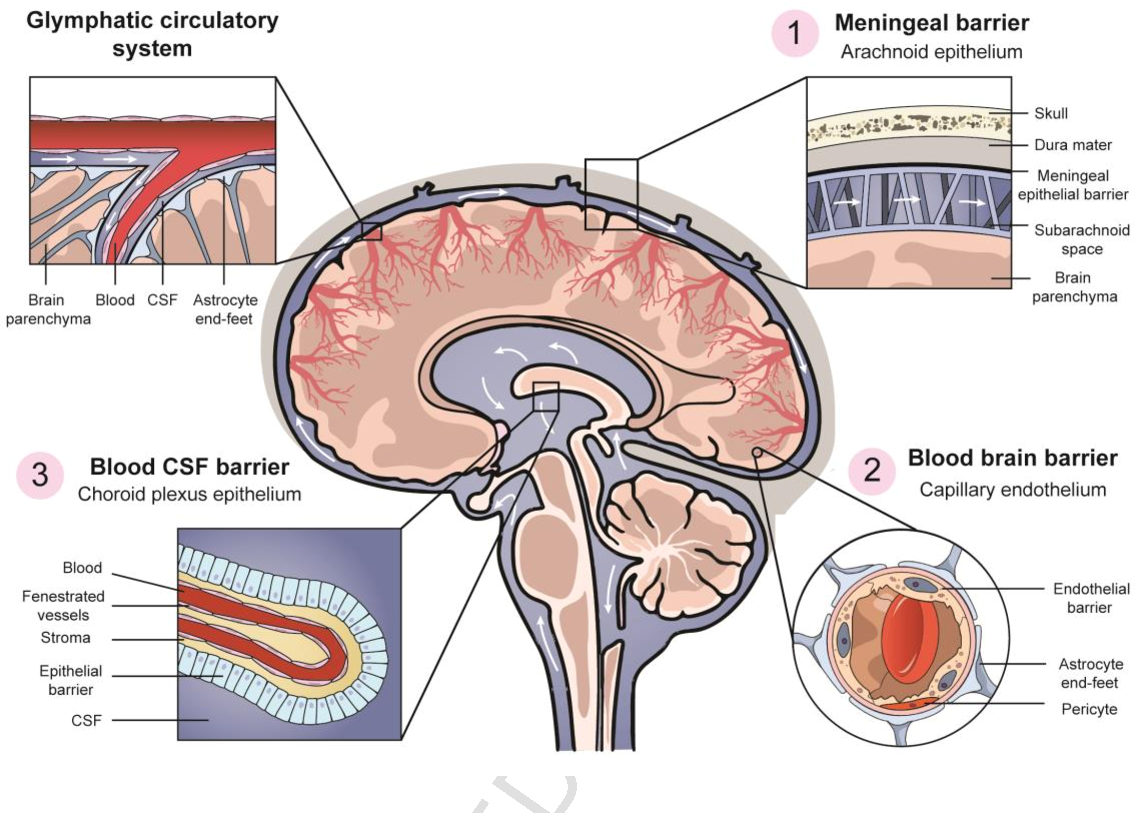
(D) In the median eminence, β1 tanycytes limit diffusion of solutes originating from the ventrally localized fenestrated capillaries into the arcuate nucleus while β2 tanycytes restrict chemical exchange between the median eminence and CSF ( Miyata, 2015 Langlet et al., 2013). Each of these CVOs possess a glia-derived, cellular barrier generated by tanycytes or tanycyte-like cells that limit further diffusion of blood-derived solutes into neighboring regions or the cerebrospinal fluid ( Ganong, 2000 Miyata, 2015). The subfornical organ, area postrema, and organum vasculosum of the lamina terminalis are the sensory CVOs, while the median eminence and neurohypophysis are the secretory CVOs. CVOs are vascularized by fenestrated capillaries and allow a small subset of neurons and glia to sense blood-derived signals or secrete hormones into the blood to regulate peripheral processes such as fluid homeostasis, osmoregulation, body temperature, energy balance, and inflammation. (B and C) These properties include (compare panel B to C): (1) a dense formation of junctional complexes that restrict paracellular diffusion of hydrophilic solutes (2) expression of efflux transporters and low rates of transcytosis that limit transcellular movement of molecules and (3) expression of selective transporters that import necessary nutrients or export wastes ( Marques et al., 2017). The choroid plexus epithelial cells possess similar properties as the ECs of the BBB. The specialized ependymal epithelial cells of the choroid plexus surround its fenestrated capillaries and filter the fluid that enters through fenestrae to generate the cerebrospinal fluid. (A) Among these regions is the choroid plexus, the structure that generates most of the cerebrospinal fluid. In these regions, a specialized glial barrier takes on the role of the endothelial BBB. There are small regions of the brain that lack an endothelial BBB and are instead vascularized by permeable fenestrated capillaries. Expression data suggest that the answer might lie in the CNS-specific enrichment of certain cytoplasmic adaptors (e.g., JACOP, MPP7) and tricellular TJ molecules such as LSR and MARVELD2 ( Daneman et al., 2010a Sohet et al., 2015). It is still unknown why CLDN5 and ZO-1 expression does not confer the same low paracellular permeability in peripheral vessels as in the CNS. Cytoplasmic proteins including ZO-1, ZO-2, ZO-3, cingulin, JACOP, MAG1, and MUPP1 aid TJ formation, binding TJs to the cytoskeleton, adherens junctions, and polarity complexes ( Umeda et al., 2004 Tietz and Engelhardt, 2015 Sawada, 2013). Other key components of TJs include claudin 12, occludin, and junctional adhesion molecules. 
ECs in peripheral vascular beds also express CLDN5, and thus its expression alone is not sufficient for barrier formation. Claudin 5 (CLDN5) is the most abundant claudin at the BBB, and Cldn5 knockout mice exhibit size-selective leakage of the BBB and die at birth ( Morita et al., 1999 Nitta et al., 2003). The composition of claudins, a family of 27 four-pass transmembrane proteins, within a TJ is thought to determine the size and charge selectivity of paracellular permeability ( Amasheh et al., 2005 Hou et al., 2006 Furuse et al., 1999). This review will discuss BBB development, regulation, and dysfunction, emphasizing important unanswered questions. The complex properties that define the BBB are often altered in disease states, and BBB dysfunction has been identified as a critical component in several neurological conditions. The interaction of ECs with different neural and immune cells is commonly referred to as the neurovascular unit (NVU Fig. The BBB tightly regulates the movement of ions, molecules, and cells between the blood and the parenchyma and is thus critical for neuronal function and protection. The BBB is not a single physical entity but rather the combined function of a series of physiological properties possessed by endothelial cells (ECs) that limit vessel permeability. Blood vessels provide the vital infrastructure for delivery of oxygen and essential nutrients throughout the body, and the term “blood–brain barrier” (BBB) is used to describe the unique characteristics of the blood vessels that vascularize the central nervous system (CNS Saunders et al., 2008 Zlokovic, 2008 Obermeier et al., 2013).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
